
Whatever the case may be, keep learning and keep explaining!H:- If so that is the Lewis dot structure for the hydride ion. We just need to get clarity in mind and focus on the small details which can help us to solve any significant issues of chemistry. Learning about various terms of chemistry just to understand Geometry of Moleculesmakes it a fun learning. I hope you got the answers and detailed explanation regarding Sulfur difluoride. So, SF2 is a polar molecule because of those differences in the electronegativity. We have the sides where there is fluorine, and then there is a side where that lone pair of electrons stays! So, because we have these different sides, this molecule is not symmetrical, and we get a negative as well as a positive side, which makes this a polar molecule. CS2 Lewis Structure, Hybridization, Polarity and Molecular Shape Here what we can see from this structure is that we have two different sides. So as the electron pairs and the fluorine spread out, we end up with this bent geometry. You can see here that we have the two lone pairs on the top and the fluorine atoms are forced down to the bottom. In short, all of these forms the geometry of the Sulfur Fluorine molecule. So according to Valence Shell Electron Pair Repulsion theory VSEPR theorythese fluorine and these two lone pairs of electrons - they are going to spread out, and when they do it, they are going to give us a molecular geometry. We can see that we have two fluorine on either side of the Sulfur and we also have two pairs of lone pair electrons. To determine whether SF2 is polar or nonpolar, first look at the Lewis structure. If we talk about the bond angles, it is 98 which is very similar to H2O. The hybridization by the central Sulfur is SP3. As I have described earlier, the two lone pairs of electron of SF2 gives it a bent shape. Whenever the canter atom has two lone pairs and two particles, the geometry is bent or angular.
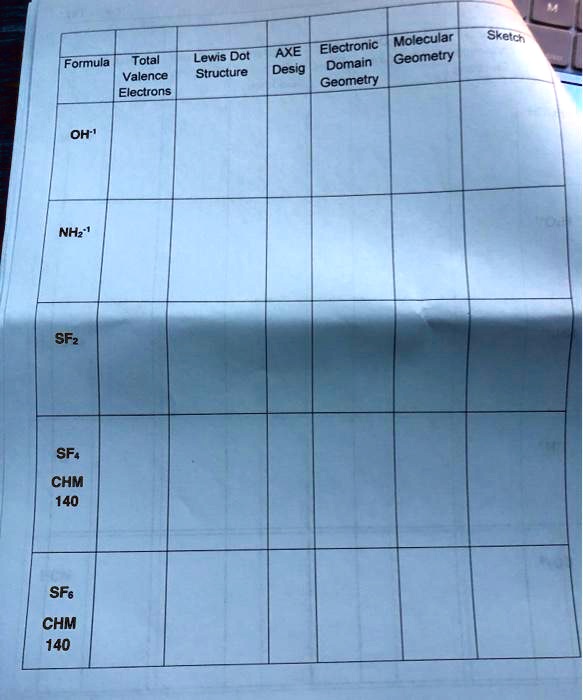
It forms one bond because it has seven valence electrons and it only needs one more to get to eight. Twenty minus Sixteen So what it tells us is that there are four electrons or two lone pairs of the central sulfur atom and fluorine. Now, when the figure is subtracted, we get four. Now we are going to subtract that sum from the highest multiple of eight but just below twenty, which is obviously sixteen.

This combination gives us the total of twenty. To know about the Sulfur Fluorine - SF2 molecule geometrythe very first thing we have to do is to add up the valence electrons.Īs you may know, Sulfur has six valence electrons, and the Fluorine has seven valence electrons. So in this article, I am going to solve all the confusions regarding of the Sulfur DiFluoride - SF2 molecular geometry. There are so many things to know about such as molecular geometry, Lewis structure, polarity, hybridization, as well as bond angles, but very little information available online. In the case of SF2, the actual bond angle is just 98 degrees.Many of my students were confused after not getting any useful information about SF2 on the internet.

Due to the lone pairs, most teachers want to hear that the bond angle is “less than 109.5 degrees”, since the lone pairs repel the bonding pairs and push the single bonds together more than they do in a tetrahedral molecule like CH4. Sulfur has two single bonds and two lone pairs around it, and this is four things, so the electron pair geometry is tetrahedral. The hybridization of the fluorine atoms is sp3 as well, since they also do not have any double or triple bonds. The sulfur atom has no double bonds, which means that no pi-bonds are needed. This gives it a VSEPR notation of AX2E2, which is angular / bent / non-linear geometry. In the end, sulfur is single-bonded to each of two fluorine atoms (this is two bonding pairs) and has two lone pairs on it as well. Sulfur is single-bonded to each of two fluorine atoms, and has two lone pairs as well. Lewis Structure of SF2 (sulfur difluoride). This means that sulfur can share ONE electron with EACH of two fluorine atoms, completing all of their octets simultaneously.
#SF2 ELECTRON DOT FORMULA FULL#
Sulfur brings 6 valence electrons with it, and so needs two extra for have a full octet.įluorine brings 7 valence electrons with it, and so needs one extra to complete its octet. They are both non-metals, so they share electrons to form covalent bonds. Sulfur difluoride (SF 2) is one sulfur atom connected to two fluorine atoms.
